 Below is the Born haber cycle for the formation of sodium chloride crystal. The energy change will be same whether it reacts in few steps or in one step. For an example, the solid sodium and chlorine gas reacts to form sodium chloride crysltal. So the total energy involved to form a ionic compound from its elements will be same whether it happened in few steps or in one step. According to this law the energy change is same for a particular reaction regardless whether the reaction undergoes in one or several steps. The calculation of lattice energy can be done by using Hess’s law (for this case it is called Born Haber cycle). Here we can see that the lattice energy of MgO is much greater than the lattice energy of NaCl. Thus +2 or -2 ions will release more lattice energy than the +1 or -1 ions. Lattice energy increases with increase of charge on the ions because of their more attractive force between them. Thus the attraction between them decreases and finally the less lattice energy released during the process. This is because with the increase of size of ions, the distance between their nuclei increases. As the size of halide increases down the group, the lattice energy decreases. Below is a graph of the lattice energy of lithium halide. a) Radius of ionsĪs the radius of ions increases, the lattice energy decrease. And lattice energy depends on two factors: size or radius of ions and charge of ions. The strength of ionic bond increases with the increase of lattice energy. That means, energy released when a cation and a anion combine together to form one mole of an ionic compound is know as lattice energy or lattice enthalpy. The energy released in this process is known as lattice energy or lattice enthalpy. After the formation of ions, they combine together to form ionic compound. The value of the Madelung constant has been calculated for all common crystal structures by summing the contributions of all the ions in the crystal lattice.Ionic compounds are more stable because of their elctrostatic force between the two opposite ions. The lattice energy of the ionic crystal is inversely proportional to the Inter-ionic distance and directly proportional to the product of charges of the ions, Madelung constant, and Born exponent. Where N A is the Avogadro constant, the number of molecules in a mole has the value 6.023×10 23 mol -1 A is the Madelung constant, which depends on the geometry of the crystal.  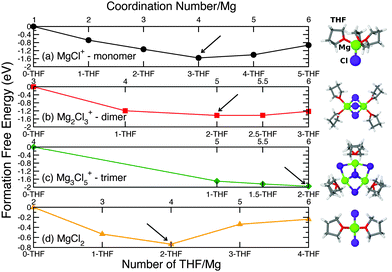 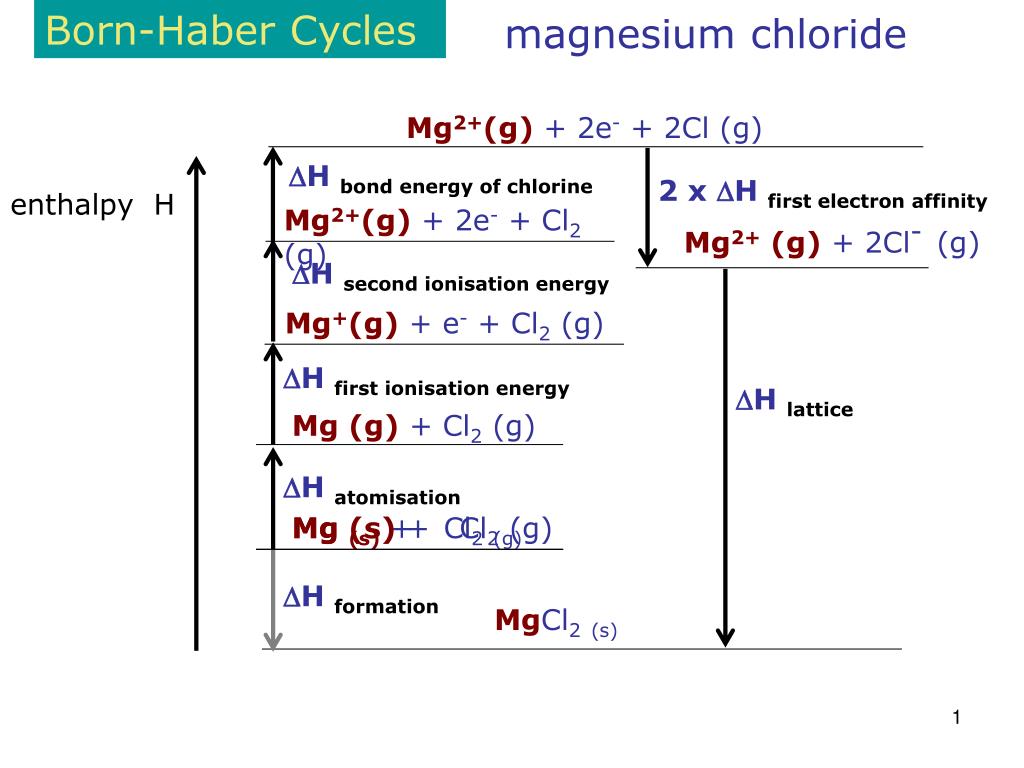
Total energy = Attractive energy + Repulsive energyįor one mole of the ionic crystal U = E total N A This is the Born Lande equation. 
M = Madelung constant, which is related to the geometry of the crystal Repulsive force where, B = constant, A portion of three-dimensional cubic lattice and its unit cell Where Z + and Z – are the charges on the positive and negative ions,Īttractive energy for a simple lattice of the crystal The ions are treated as point charges, and the electrostatic energy E between two ions of opposite charge is calculated. Theoretical values for lattice energy may be calculated. Lattice energies cannot be measured directly, but experimental values are obtained from thermodynamics data using the Born Haber cycle. Lattice energy is defined as the energy released in the process when the constituent ions are placed in their respective positions in the crystal lattice or, the amount of energy required to separate the solid ionic crystal into its constituent ions. Schematic representation of lattice energy at inter-ionic distance r o For sodium chloride, the lattice energy, U, is equal to the enthalpy change for the reaction. The lattice energy (U) of a crystal is the energy that evolved when one gram of the crystal is formed from gaseous ions. 
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
